JCA update available
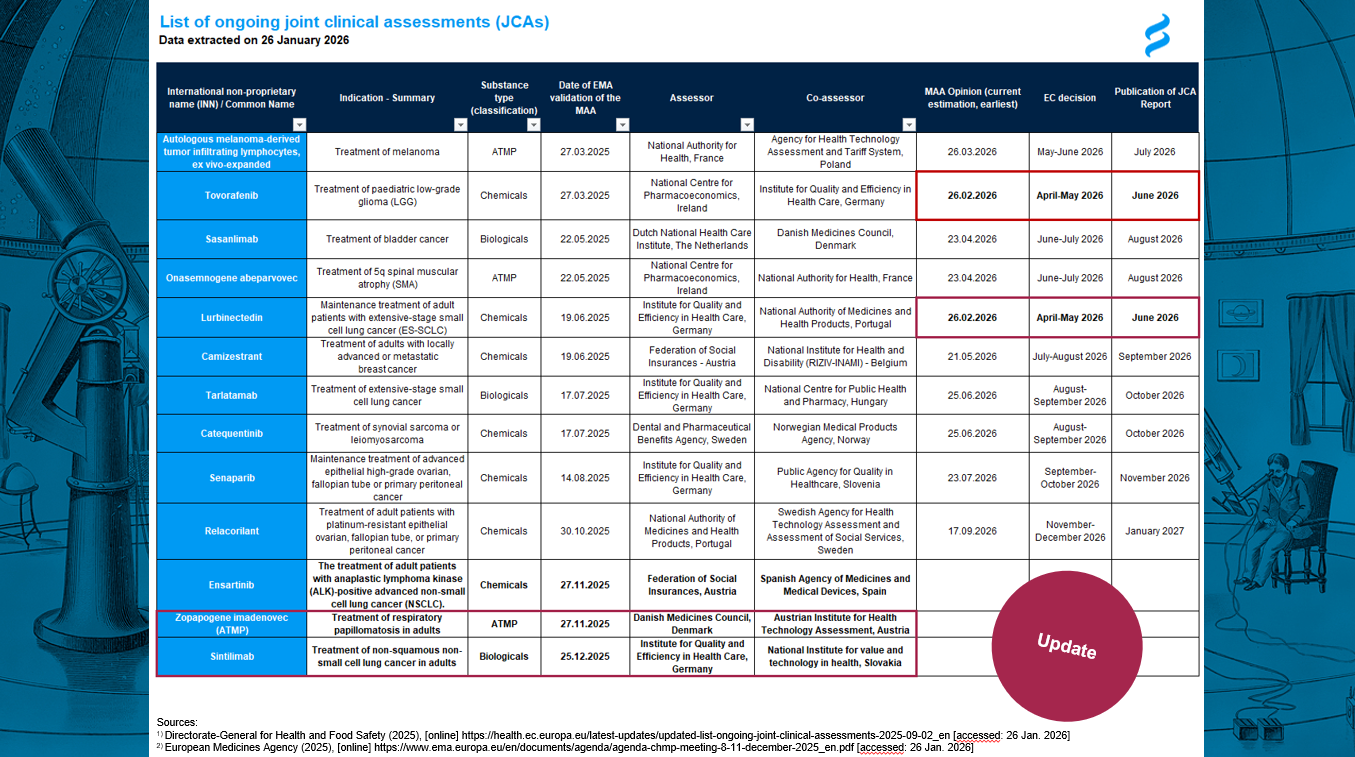
📢 JCA update available
💡 We provide you with the latest regulatory update from the EMA, announced last week.
🟥 Find the updates in the red frames. 🟥
Key findings:
🔹 On 26 March 2026, the CHMP adopted positive opinions for PharmaMar’s Zepzelca (lurbinectedin) and Amgen’s Imdylltra (tarlatamab), both indicated for adults with extensive-stage small cell lung cancer, ES-SCLC, addressing maintenance therapy following first-line treatment and relapsed disease, respectively. JCA reports are expected around early July 2026.
🔹 Autologous melanoma-derived tumor infiltrating lymphocytes, ex vivo-expanded received the D180 LoOI.
🔹 AstraZeneca’s camizestrant, for the treatment of adults with locally advanced or metastatic breast cancer, received the D180 LoOI.
🔭 Take a look at the analysis in our observatory.
Disclaimer: Our analysis is compiled using only publicly available information and assumes a standard EMA timeline of 3 months clock stop d120 and 1-month d180, except where clock stop extensions are agreed with EMA. Subject to change without notice.
We do not guarantee the accuracy of the information provided here.