A major step forward for patients
🚀 Ipsen’s tovorafenib for paediatric low-grade glioma received a positive CHMP opinion on 26 February, recommending a conditional marketing authorisation.
A major step forward for patients – and, from a Market Access perspective, a very telling example of how EMA and EU HTA timelines are intertwined.
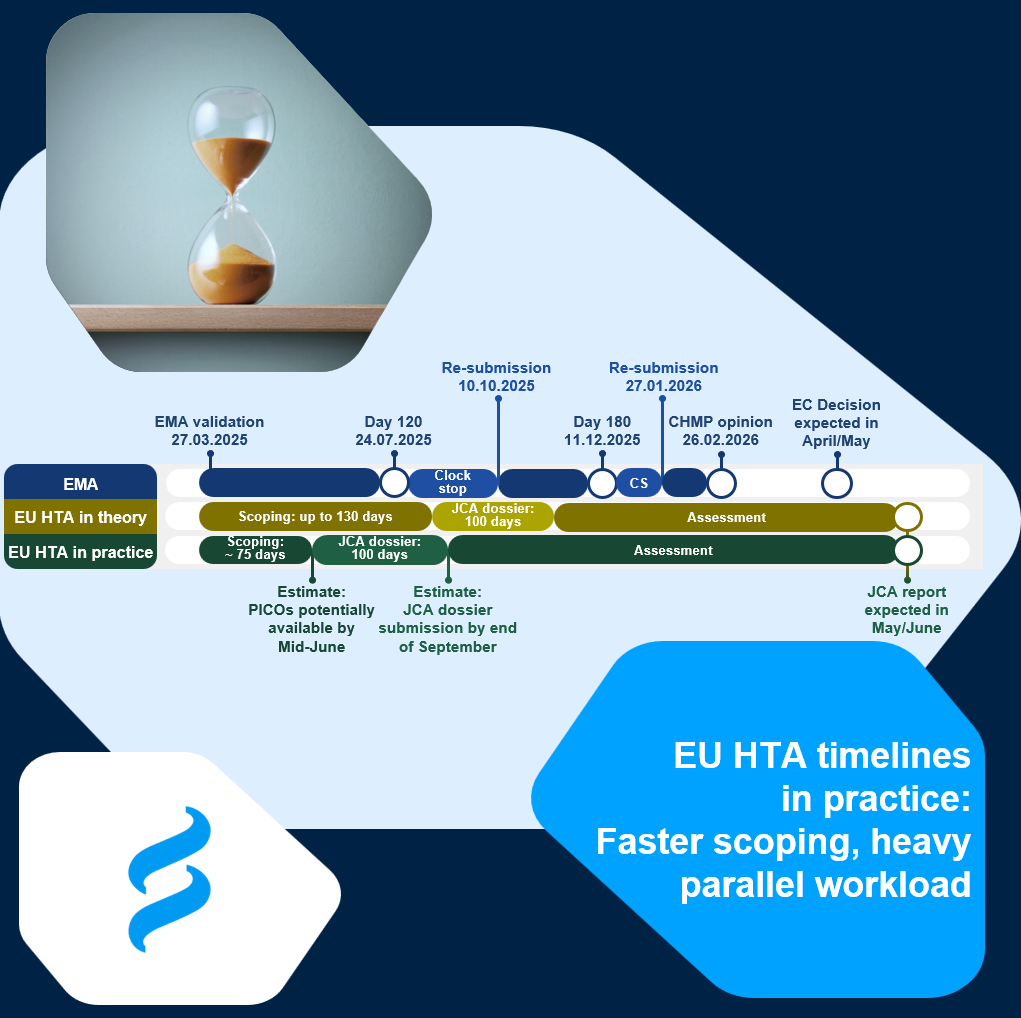
📊 We mapped the EMA procedure (Day 120 LoQ on 24 July 2025; Day 180 LoOI on 11 December 2025) against the EU HTA timelines – once as per the Regulation, and once based on what we are currently seeing in practice.
A few key takeaways:
⏱️ Scoping is moving faster than expected
While the Regulation foresees up to 130 days, we are currently observing an average scoping duration of ~75 days. The Coordination Group is clearly gaining speed here.
⚖️ The double workload is real
For the pharmaceutical company, the JCA dossier needs to be progressed and finalised in parallel with responding to the Day 120 questions. That overlap creates significant operational pressure.
📌 The longest phase remains the assessment
Within the EU HTA process, the assessment phase – under the responsibility of the Coordination Group –represents the largest time block.
Curious to hear how others are experiencing the practical alignment of EMA and EU HTA timelines in ongoing procedures.