12.03.2026
Update on sasanlimab and EU HTA
📢 Update on sasanlimab and EU HTA
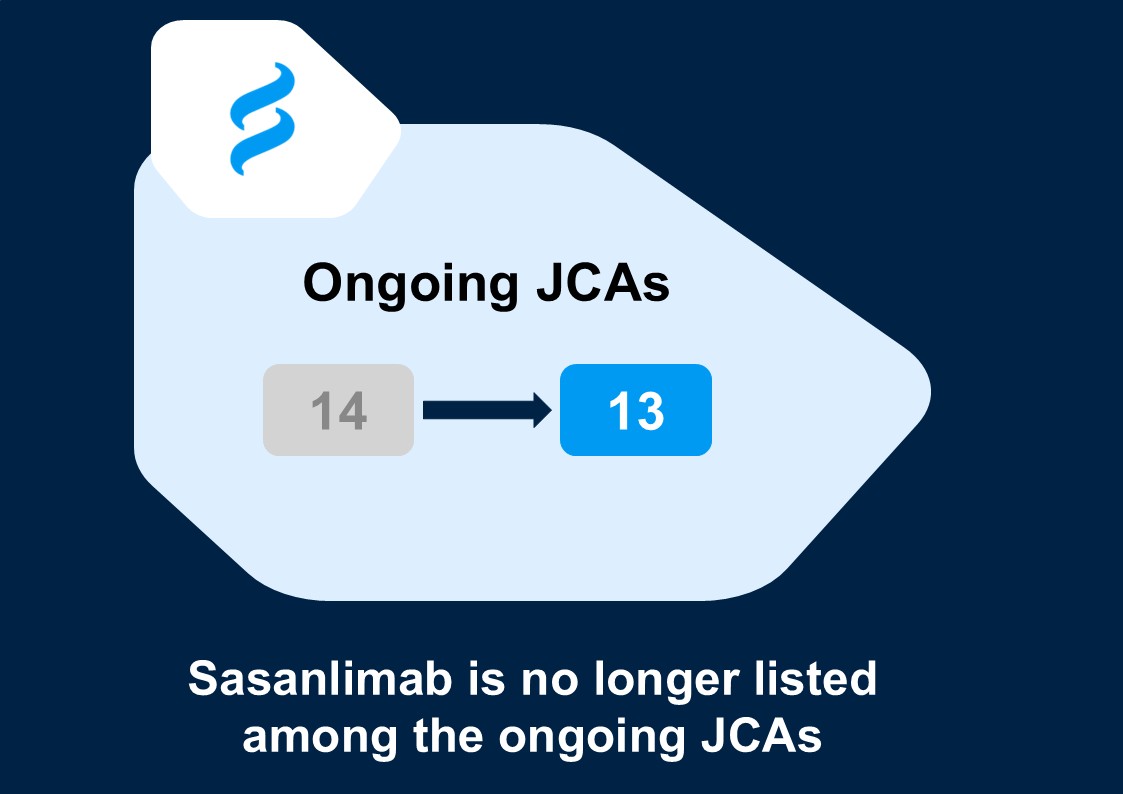
Pfizer has withdrawn the EU marketing authorisation application for sasanlimab, which in turn means that the associated Joint Clinical Assessment (JCA) has been discontinued. Sasanlimab is no longer listed among the ongoing JCAs. ❌
This makes sasanlimab the first JCA to be formally discontinued under the EU HTA framework, reducing the number of ongoing JCAs from 14 to 13. 📉
🔎 It will be interesting to see what information, if any, will be published from this discontinued procedure and what this implies for transparency and learning effects in future JCAs.